This year Impeto Medical celebrates its
15th anniversary
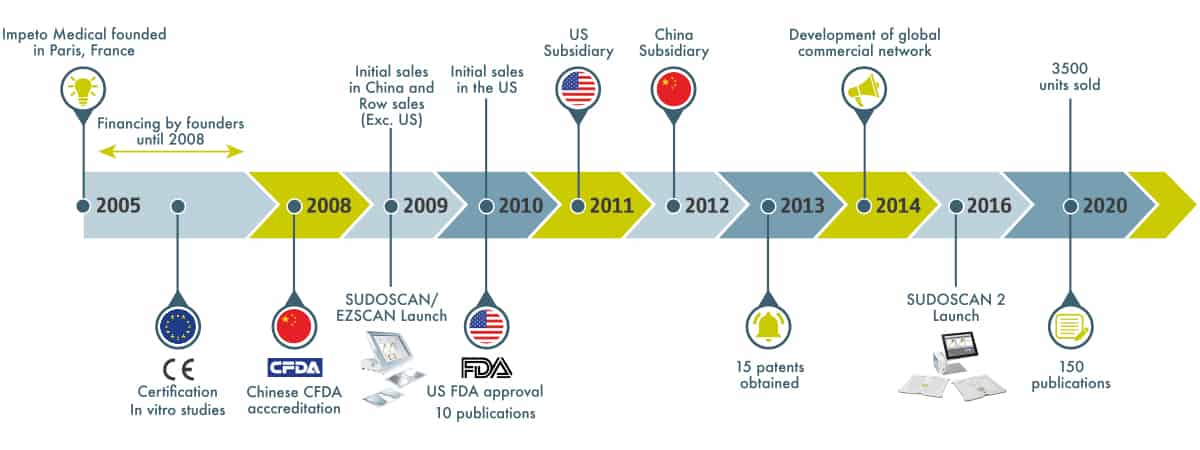
Over the past 15 years, the company has made remarkable progress in R&D and sales, with more than 3,500 units sold and several million tests per year in 40 countries around the world, more than 150 articles published in peer-reviewed journals and 15 patents filed.
Considering the current and future medical and technological challenges, SUDOSCAN’s success is still ongoing, contributing to improve patient management and quality of life.
It all started with the ingenious ideas of the founders in 2005 and the creation of the French headquarters in Paris. Then the first product prototypes were patented and obtained the CE mark. In vitro studies were milestones to support registration in China and the USA.
The first SUDOSCAN generation was launched in 2009 as a unique non-invasive tool to assess small fiber neuropathies.
Bellow evolvement steps of the company and its products.
Click on the image below to zoom
This amazing technology has been recognized within the medical communities thanks to hundreds of independent clinical studies performed in major areas such as prediabetes, diabetes, neurological diseases and oncology. As of today, SUDOSCAN is mentioned in international medical guidelines to manage peripheral neuropathy and diabetes complications:
French Health Authority (HAS), National Protocol for Diagnosis and Care for Familial Amyloidosis Neuropathy.
German Diabetes Society – Practice recommendation for Diabetic Neuropathy.
Polish Diabetes Association – Guidelines on the management of diabetic patients.
ALAD, Latin-America Diabetes Association – Expert consensus for management of diabetic neuropathy.
New England Journal of Medicine – Diabetic Sensory and Motor Neuropathy (Clinical Practice).
The Lancet Diabetes and Endocrinology – Advances in diagnosis and strategies for screening diabetic polyneuropathy.
SUDOSCAN is a French-made medical device with the highest quality requirements. Since 2019, Impeto Medical’s manufacturing is managed by a new large partner Alliansys, based in the Normandie region.